🦴 Orthopedic implants are among the most advanced products in modern medical manufacturing. From hip replacements and spinal cages to trauma plates and fixation screws, these devices must withstand years of stress inside the human body while maintaining precision, safety, and long-term reliability.
For OEM manufacturers, hospital procurement teams, distributors, and orthopedic brands, understanding the different types of orthopedic implants, material selection strategies, and production technologies is critical for making informed commercial and clinical decisions.
In this guide, we explore the major implant categories, the most widely used orthopedic implant materials, and the manufacturing technologies shaping the future of orthopedic surgery.
For related topics, explore our cluster articles: Hip, Knee and Shoulder Implants, Spinal Implants, Trauma Implants, Orthopedic Implant Materials, and 3D Printed Orthopedic Implants.
For industry trends and demand forecasts, see our Global Orthopedic Devices Market guide.
What Are Orthopedic Implants?
🔬 Orthopedic implants are medical devices surgically placed into the musculoskeletal system to restore function, stabilize injuries, or replace damaged anatomy. They are commonly used in joint reconstruction, fracture fixation, spinal fusion, and complex bone reconstruction procedures.
These implants generally serve four primary clinical purposes:
- Replacement — replacing damaged joints such as hips or knees
- Fixation — stabilizing fractured bones during healing
- Fusion — permanently joining vertebral segments
- Reconstruction — restoring bone loss caused by trauma or tumors
Regulatory classification also plays a major role in commercialization. The FDA’s medical device classification database categorizes most orthopedic implants as Class II or Class III devices depending on complexity and risk profile.
Classification by Anatomical Application
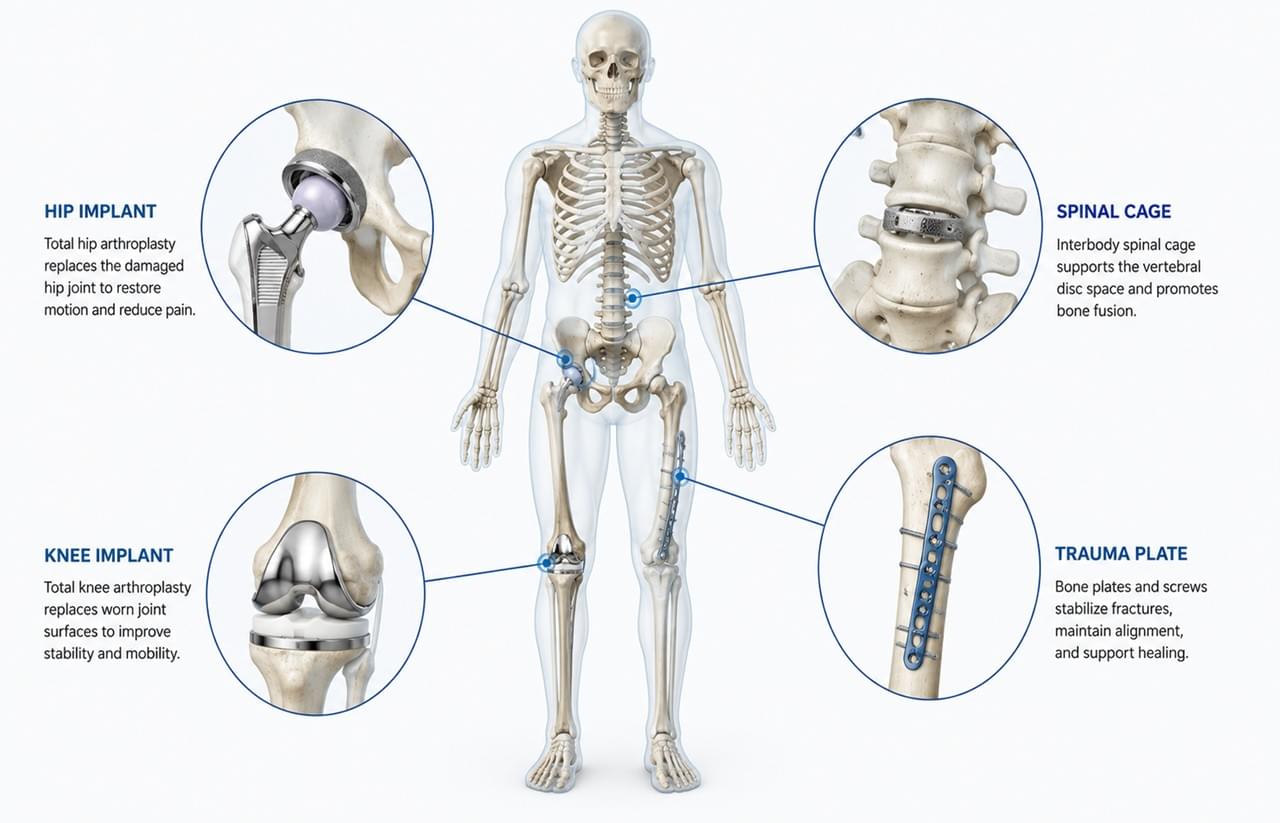
🦿 Joint Reconstruction Orthopedic Implants
Joint reconstruction represents the largest segment of the global orthopedic implants market.
Total Hip Arthroplasty (THA) Systems
Modern hip systems typically include:
- Acetabular cup and liner
- Femoral stem
- Femoral head
- Bearing couples such as ceramic-on-polyethylene or ceramic-on-ceramic
Titanium alloys and cobalt-chrome alloys dominate this category because of their strength and wear resistance.
Total Knee Arthroplasty (TKA) Systems
Knee replacement systems generally include:
- Femoral component
- Tibial tray and insert
- Patellar component
Design variations such as cruciate-retaining and posterior-stabilized systems help surgeons adapt implants to different patient conditions.
Shoulder Arthroplasty Systems
Shoulder implants include both anatomical and reverse shoulder systems designed for arthritis, trauma, or rotator cuff deficiency.
For deeper comparison, read: Hip, Knee and Shoulder Implants: Key Differences and Applications.
🧠 Spinal Orthopedic Implants
Spinal systems are among the fastest-growing categories of orthopedic implants due to increasing degenerative spine conditions worldwide.
Common Spinal Implant Systems
- Interbody fusion cages
- Pedicle screw and rod systems
- Cervical plates
- Artificial disc replacements
- Dynamic stabilization systems
Materials such as PEEK and porous titanium are widely used because they support imaging visibility and bone integration.
Explore more here: Spinal Implants: Design Principles and Clinical Applications.
⚙️ Trauma Orthopedic Implants
Trauma implants are designed to stabilize fractures and support bone healing.
Common Trauma Implant Types
- Locking plates
- Intramedullary nails
- Cannulated screws
- Kirschner wires (K-wires)
- Compression screws
Different fracture patterns require different fixation strategies, making product variety essential in trauma portfolios.
See also: Trauma Implants: Plates, Screws and Intramedullary Nails.
Orthopedic Implant Materials Explained

Choosing the right orthopedic implant materials is one of the most important engineering decisions in implant design.
🔹 Titanium Alloys (Ti-6Al-4V)
Titanium remains the leading material for structural orthopedic implants because it offers:
- Excellent biocompatibility
- High corrosion resistance
- Strong osseointegration performance
- Lower elastic modulus compared to cobalt-chrome
- MRI compatibility in many applications
Titanium is commonly used in hip stems, spinal screws, trauma plates, and porous implant systems.
🔹 Cobalt-Chrome Alloys (CoCr)
Cobalt-chrome alloys are preferred in high-wear applications.
Advantages of CoCr
- Exceptional hardness
- Superior wear resistance
- High fatigue strength
- Excellent polishing capability for articulating surfaces
These materials are widely used in femoral heads and knee femoral components.
🔹 PEEK (Polyether Ether Ketone)
PEEK has become increasingly important in spinal implant applications.
Why PEEK Is Popular
- Radiolucent under imaging
- Elastic modulus similar to cortical bone
- Lightweight and chemically stable
- Excellent fatigue resistance
PEEK is widely used in spinal cages and vertebral body replacement systems.
See more: Orthopedic Implant Materials: Titanium, PEEK, Stainless Steel & More.
🔹 UHMWPE and XLPE
Ultra-high molecular weight polyethylene is primarily used in joint bearing surfaces.
Modern cross-linked polyethylene variants significantly reduce wear and improve implant longevity, especially in hip and knee replacements.
🔹 Stainless Steel (316L)
Although less common in permanent implants today, stainless steel still appears in cost-sensitive trauma systems.
Typical Applications
- Temporary fixation systems
- Fracture plates
- Entry-level trauma implants
Its lower cost remains attractive in developing healthcare markets.
Orthopedic Implant Manufacturing Technologies

Modern orthopedic implant manufacturing technologies combine precision engineering with advanced materials science.
🏭 CNC Machining
CNC machining remains the dominant production method for many orthopedic implants.
Key Advantages
- Tight dimensional tolerances
- Complex geometries
- Excellent repeatability
- High production consistency
Five-axis CNC systems are widely used for titanium and cobalt-chrome implant manufacturing.
🔥 Casting and Forging
Investment Casting
Used mainly for cobalt-chrome components with complex geometries.
Forging
Forged implants often demonstrate better fatigue performance than cast components, making forging ideal for high-load implants such as femoral stems.
🖨️ Additive Manufacturing (3D Printing)
Additive manufacturing is rapidly transforming orthopedic implant manufacturing technologies.
Key Benefits of 3D Printing
- Porous lattice structures for bone ingrowth
- Patient-specific implants
- Reduced implant weight
- Faster prototyping and customization
Learn more here: 3D Printed Orthopedic Implants: Benefits and Applications.
🧪 Surface Treatment Technologies
Surface engineering directly influences implant integration and long-term performance.
Common Surface Treatments
- Porous coatings
- Hydroxyapatite (HA) coatings
- Titanium anodization
- Antimicrobial coatings
These treatments help improve osseointegration, reduce infection risks, and enhance implant durability.
Patient-Specific Orthopedic Implants
📈 Personalized medicine is creating strong demand for patient-specific orthopedic implants.
Customized implants are often used in:
- Complex revision surgeries
- Tumor reconstruction
- Severe anatomical deformities
- Personalized spinal procedures
Most patient-specific systems are produced using advanced imaging combined with additive manufacturing or precision CNC machining.
Quality and Regulatory Requirements
Quality control is critical in orthopedic manufacturing because implant failure can have severe clinical consequences.
✅ Manufacturing Quality Standards
Manufacturers typically require:
- ISO 13485 certification
- Process validation protocols
- Mechanical fatigue testing
- Material traceability systems
- Dimensional inspection procedures
🌍 Global Regulatory Pathways
Major regulatory frameworks include:
- FDA 510(k) and PMA (United States)
- EU MDR CE marking (Europe)
- NMPA registration (China)
- PMDA approval (Japan)
For more details, visit: Orthopedic Device Regulatory Compliance.
FAQ About Orthopedic Implants
What are the most commonly used orthopedic implant materials?
Titanium alloys are the most widely used structural materials because they combine strength, corrosion resistance, and biocompatibility.
How long do orthopedic implants last?
Many modern joint replacement systems are designed to function for more than 20 years under normal clinical conditions.
According to the AAOS, many total knee replacements remain functional after two decades.
Are 3D-printed orthopedic implants approved?
Yes. Multiple FDA-cleared 3D-printed orthopedic implants are already commercially available worldwide.
What certifications do implant manufacturers need?
ISO 13485 certification is considered essential for orthopedic implant manufacturers, along with country-specific regulatory approvals.
🚀 Orthopedic implants remain one of the most technically demanding and innovation-driven sectors in medical device manufacturing. As materials science, additive manufacturing, and precision engineering continue evolving, implant performance and personalization are improving rapidly.
For manufacturers, distributors, and procurement specialists, understanding implant classifications, material properties, and production technologies is essential for making smarter long-term decisions in a highly competitive market.
Looking for orthopedic implant OEM or contract manufacturing support? Contact our team to discuss production capabilities, certifications, and partnership opportunities.
Medical Disclaimer
This article is intended for informational and educational purposes only for orthopedic industry professionals. It does not constitute medical, clinical, or regulatory advice. Always verify technical specifications and compliance requirements with qualified experts and regulatory authorities.